There’s no organ more fundamental to our sense of self and identity than the brain. Since the brain was first considered the seat of consciousness in ancient Greece, scientists and philosophers have been on an endless quest to crack the mysteries within our own skulls. Almost 2500 years later, we can map the human brain down to nanoscale resolution. But even with this level of technological insight, we have yet to fully understand a simple worm’s brain of only 302 neurons, let alone a human brain of almost 100 billion neurons. This complexity unfortunately not only resides within understanding normal brain physiology, but also across aging and the pathological conditions of neurodegeneration. Billions and billions of dollars have been spent on research on Alzheimer’s and other common forms of neurodegeneration, resulting in some progress, including the approval of two recent drugs, but it’s clear much more work remains to be done. Tackling brain aging will be a monumental task that will need new strategies beyond the one-drug one-target paradigm that has been commonplace in the past.
So, what are some of the innovative strategies ongoing and how far along are they? I’ll start with one that seems far fetched but is surprisingly well-developed – cell transplantation. As a disclaimer, these strategies are early in development and not ready for commercial prime time. Before we dive in, I’ll provide some context about the brain.
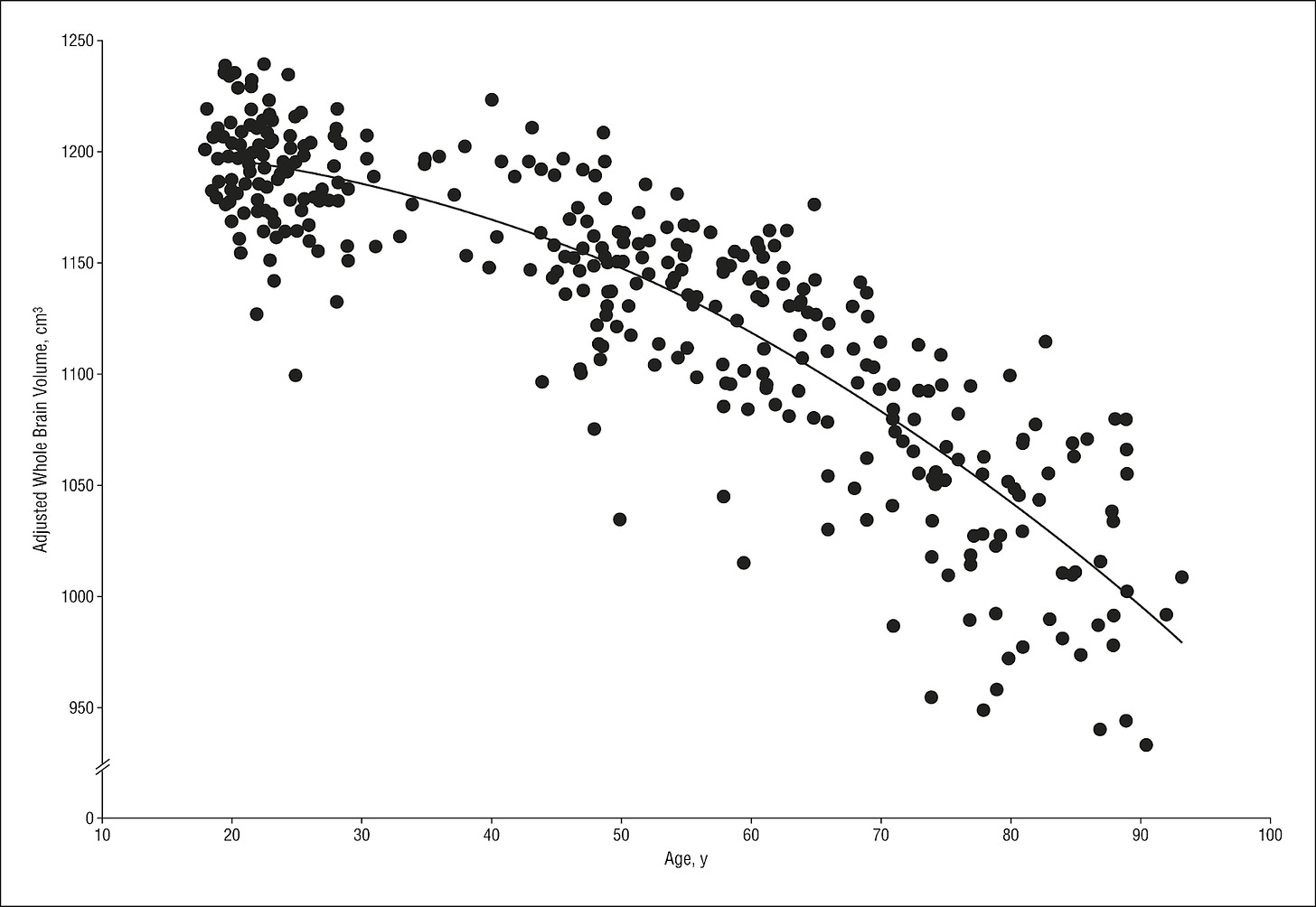
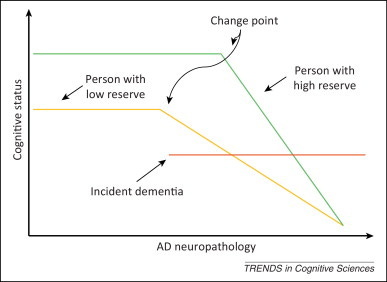
The brain is remarkably plastic throughout our lives while simultaneously experiencing degeneration with age. For instance, synapses (the structures between neurons where electrical and chemical signals are transmitted) are dynamic environments. Their strength and protein composition ebb and flow, underpinning our ability to learn. Yet unfortunately, we lose synapses as we age. Additionally, if one part of the brain is not being used (due to injury or loss of vision or hearing or a surgical procedure), that region is amazingly rewired and put to use for a different function. Yet with increasing age, our normal physiological brain connectivity and wiring deteriorate. Furthermore, as adults we can still make a small number of new neurons in the hippocampus through neurogenesis to potentially play a role in human cognition. Yet beginning in early middle age, we have significant brain atrophy and volume loss, which is further accentuated in neurodegenerative states. It is thought that there is critical point of neuronal loss where once enough neurons are gone, cognitive decline will set in. It’s like being on an inflatable lifeboat with a hole, you’ll have enough air to float for a while, but eventually you’ll eventually sink. This is visible in neurodegeneration stages where the cognitive function worsens as neuronal loss increases. In other words, having more neurons may delay this time to symptoms.
MRI scans of brain volume in 362 healthy adults across lifespan
So, if we lose brain volume with age, why not just add more? This is the philosophy behind cell transplantation strategies in neurodegeneration. They directly target the ultimate pathological feature by transplanting cells into diseased regions of the brain where there is a large amount of neuronal degradation. These cells ultimately replace the dead neurons and integrate into the already existing neural circuits. That’s what has been in development for Parkinson’s disease for nearly 45 years. Attempts at neural transplantation have been made since 1890 but were stifled until our scientific understanding grew after the 1960s. Initially studies with grafted fetal tissue in the 1980s showed promise by demonstrating these cells integrate into existing neural circuitry where they can function normally to release dopamine and ultimately survive even up to a staggering 24 years later. Some Parkinson’s patients were even able to taper down their medication level. This level of recovery is simply unheard of in the neurodegenerative space. Other neurodegenerative diseases such as Huntington’s disease also showed promise with these fetal grafts. However, fetal tissue is an unsustainable resource due to its ethical, legal & sourcing limitations. In addition, there are cases of fetal tissue generating erratic movements in patients not to mention the issues regarding immune suppression required to avoid rejection.
Graphical representation of how cognitive reserve can help delay time to dementia onset.
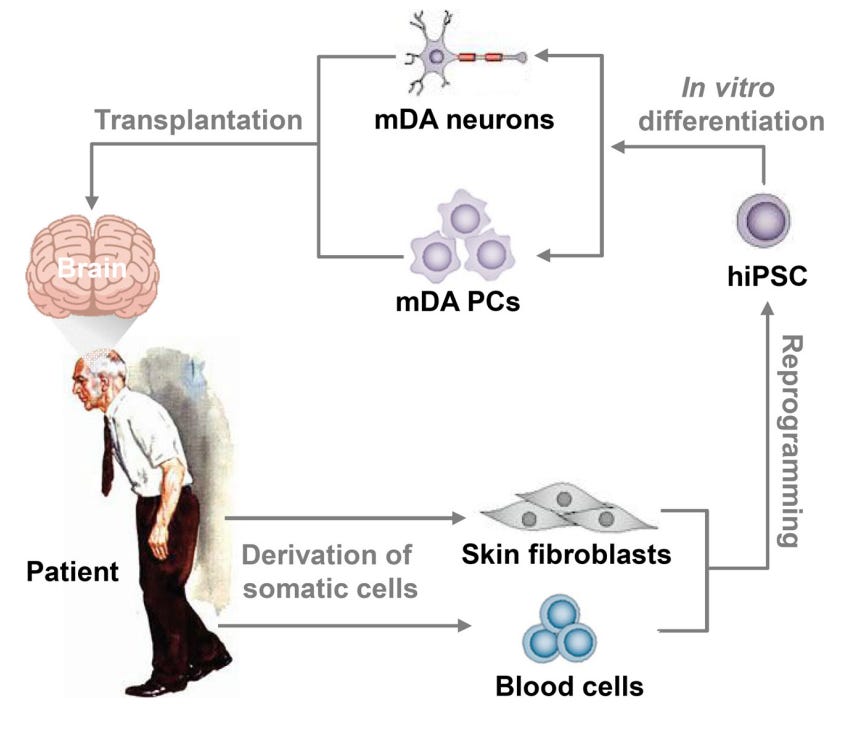
Subsequent work had propelled this field forward by searching for a more accessible and engineerable source of cells. The Nobel Prize-winning discovery of the ability to turn regular somatic cells into induced pluripotent stem cells (iPSCs) by Shinya Yamanaka, revamped this field where nearly every cell transplantation strategy now utilizes iPSCs. This enables patients to be their own source of cells. A patient’s skin cells can be turned into their own neurons or glial cells, which solves the issues of accessibility and immune rejection. This process of reprogramming into iPSCs reverses aspects of molecular aging in a cell to provide young new neurons to patients. This in combination with developments in genetic engineering in the past years can allow modification of these grown neurons to correct genetic disorders, such as the case in Huntington’s disease where genetic repeats in the Huntington protein cause the disease. Indeed, iPSC cell transplants have shown promise in animal models, including primates.
The logic behind cell transplantation for Parkinson’s disease from human iPSC (hiPSC) to midbrain dopamine (mDA) neurons and progenitor cells (PCs)
These advancements and decades of fundamental animal preclinical work has pushed this approach closer to the bedside. Private companies have started investing in this technology with the likes of Neurona Therapeutics, Aspen, Kenai, BlueRock Therapeutics. This approach is in early development with phase 1 or 2 human trials showing that they have reasonable safety and tolerability with larger efficacy studies underway. While the approaches & diseases by these companies vary, they can be separated along the allogenic (Neurona, Kenai, BlueRock Therapeutics) or autologous (Aspen) path. Allogenic focuses upon developing an off-the-shelf product that is usable for every patient but may require immune suppression or modifications to make the cell invisible to the immune system. Whereas autologous is a patient-specific personalized approach developed from a patient’s own cells. There is immense potential in these strategies to add meaningful years to a neurodegenerative patient’s life. But of course there is still much work to be done to understand the long-term stability and impacts of this approach, even if it is only a few million cells being added. For instance, epilepsy patients can have a few centimeters of their brain (hundreds of millions of neurons) removed as a therapy for seizures and there can be profound shifts in one’s identity. So even if it is a small number of cells being added, it may still have a large impact on a person’s brain.
Neuronal cell replacement is well-justified in late neurodegeneration where quality of life & basic cognitive function can deteriorate progressively. Even in early mild cognitive impairment, an invasive procedure like this may have a tremendous impact on delaying the onset of cognitive impairment. However for general aging, even if an increase of neuron number could be helpful for preventing dementia and cognitive dysfunction, intensive neurosurgery and infusing millions of new neurons into a patient’s brain may be a risky far cry for healthy non-demented elderly.
However beyond replacing lost neurons, there are other transplantation approaches that can also be applied to general brain aging. Sana Biotechnology and the affiliated Steven Goldman from University of Rochester have been investigating infusing glial progenitor cells into the brain. Glial cells are supportive non-neuronal cells in the brain. These precursor cells are not yet committed to what type of cell they will become but will eventually differentiate into mature oligodendrocytes and astrocytes. Astrocytes are important for providing energetic & metabolic support and are critical factors in the niche, regulating synaptic activity and the blood-brain barrier. Oligodendrocytes are important for maintaining and creating myelin in the brain, which is critical for electrical signal transmission between neurons. This study by Dr. Steven Goldman is beyond fascinating in the approach and insights it provides. They inject healthy human glial progenitor cells into a mouse that has diseased Huntington’s human glial cells. The healthy glial cells ultimately outcompete and actively hunt down the diseased Huntington’s glial cells. The glial cells can be observed to migrate up to 2 millimeters away from injection site. This replacement can extend survival of Huntington’s mice, improve motor dysfunction, as well as other disease phenotypes. Dr. Goldman has also shown these progenitor cells can actively remyelinate the brain and can thus serve as an approach to recover lost myelin in diseases or in aging. Sana Biotechnology is currently looking to apply this technology to Huntington’s disease and multiple sclerosis. But what about aging? Well, Dr. Goldman goes on to show that young glial progenitor cells can actively spread and replace old glial cells in a mouse. As dysfunctional glial cells are a major player in brain aging, this approach can be a powerful strategy to turn back the clock on an aging brain. This work is still in preclinical development but will be beyond fascinating to watch grow.
Beyond neurons and glia, there are other cell transplantation approaches that have shown some promise. You would think it would be pretty far-fetched that injecting fat cells into the brain would be helpful, but surprisingly transplanting mesenchymal stem cells has shown some promise. Mesenchymal stem cells are derived from a few sources, like the bone marrow or from fat, and can turn into bone, muscle or fat cells. In vitro work has shown that these mesenchymal stem cells secrete factors that can reduce senescent cell burden, reduce oxidative damage and improve cognition in a mouse model with high amount of senescence. Other animal work has shown that injecting these stem cells directly into the brain of an Alzheimer’s mouse model can reduce inflammation, induce autophagy and improve cognition but does not impact amyloid or neurodegeneration. A recent study expanded upon this body of work to investigate transplantation in an aging mouse model and found improved cognition and increased levels of a critical brain factor called brain-derived neurotrophic factor (BDNF). Animal trials are one thing altogether, but human clinical trials are also underway with this approach. Longeveron’s approach of an allogenic intravenous injection has shown benefits in a phase 2a trial by reducing inflammation & brain volume loss while also improving cognition. A company called NeuroGenesis is investigating injected autologous bone marrow stem cells into the spinal cord or directly into the blood for multiple sclerosis. These cells apparently are present for up to three months. In a phase 2a trial, they have shown the treated group were less likely to functionally deteriorate and even had improvements of motor and cognitive function. They are now expanding in their phase 2b trials. Brainstorm Cell Therapeutics is another company that is quite ahead in this space with another autologous bone marrow approach with a phase 3a trial for ALS and a phase 2 in multiple sclerosis where they saw improvements in biomarkers of both diseases. Another company, Regeneration Biomedical, is amid a Phase 1 clinical trial for autologous mesenchymal stem cells isolated from a patient’s fat and directly infused into the lateral ventricles of the brain. These sorts of approaches may also apply for age-related myelin loss and for targeting aging-related neuroinflammation and are thus worth keeping an eye on.
All in all, while we have made remarkable strides in understanding and mapping the human brain, the complexity of this organ will continue to challenge scientists for decades to come. While we have so far only seen modest success in targeting many diseases of the brain, there may be some hope with cell transplantation by potentially restoring lost neurons or glia and offering personalized treatments.
Table References (by row)